Publications are the currency of breakthrough science
Our investigators are among the most prolific in the life sciences
Publications
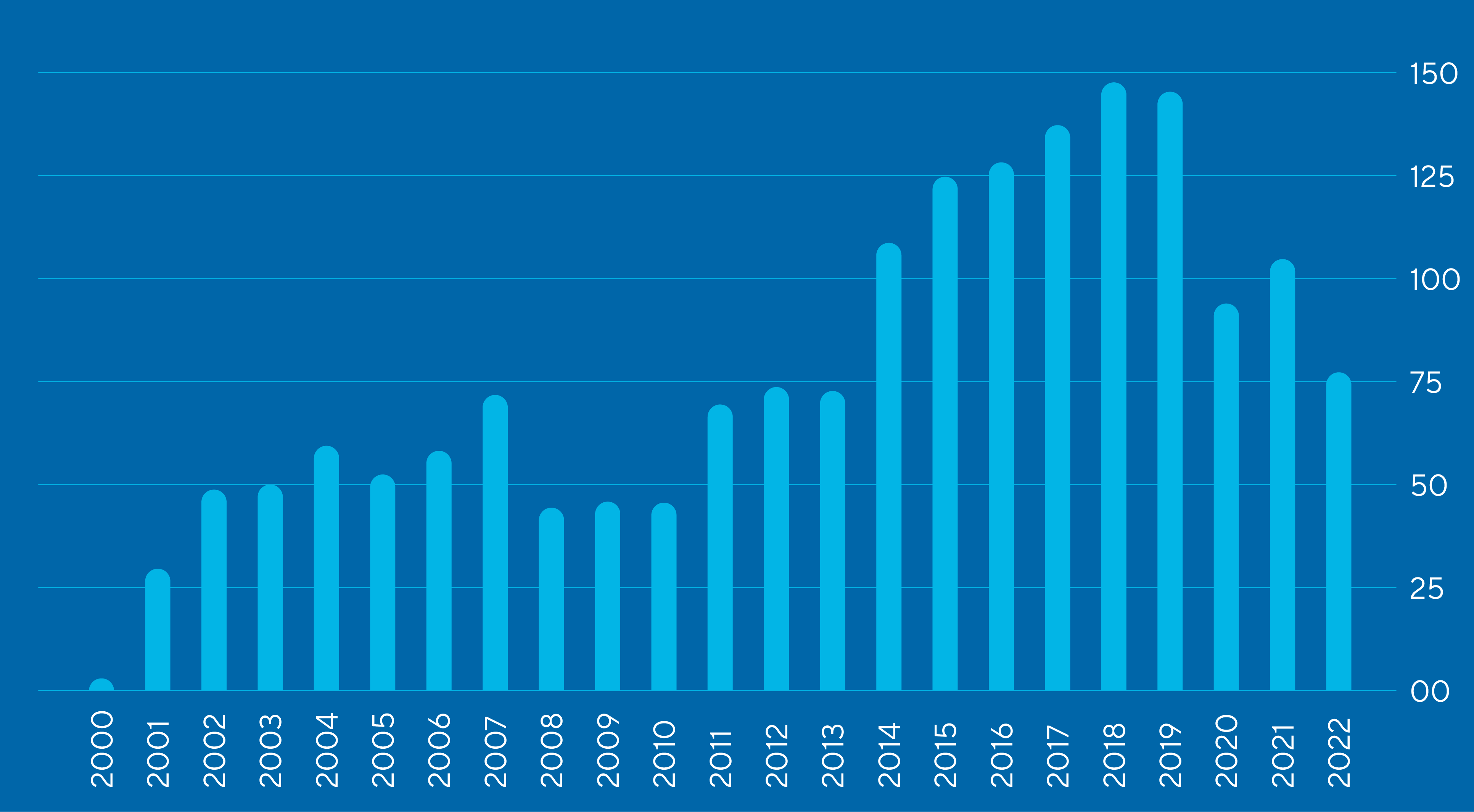

Our investigators are among the most prolific in the life sciences

Notifications